Tetramethylammonium Hydroxide (TMAH) manufactured by SACHEM is of the highest quality available worldwide. As a quaternary ammonium compound (Quat), TMAH plays a crucial role in etching solutions and is widely used as a photoresist developer in the manufacturing of integrated circuits, flat panel displays, printed circuit boards, capacitors, sensors, and other high-purity chemical applications. The molecular formula is N(CH₃)₄⁺ OH⁻, and it is commonly manufactured as an aqueous solution, in methanol, or in propelyne glycol. With decades of expertise, SACHEM is one of the oldest and most trusted tetramethylammonium hydroxide suppliers for semiconductor and industrial applications.
Tetramethylammonium Hydroxide Benefits:
SACHEM’s Tetramethylammonium Hydroxide (TMAH) offers unmatched reliability and quality for high-volume production. With manufacturing facilities in America and Asia, we provide global shipping to meet your needs wherever you are.
- Metal-Free Composition: Contains no metal.
- No Residual on Heating: Leaves no residue, maintaining the integrity of your processes.
- Ultra-Low Contaminant Levels: Exceptional purity for high-tech manufacturing.
- Application-Specific Grades: Tailored grades available to suit various industry needs.
- Custom Specifications: We work with you to create TMAH solutions that meet your unique requirements.
- Consistent Product Quality: Dependable performance for every batch.
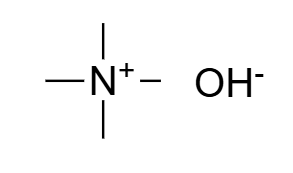
SACHEM’s TMAH: High-Purity for Semiconductor & Industrial Applications:
- Advanced Ceramics
- Electrolytes
- Catalyst
- Trans-esterification catalyst
- Photoresist developer
- Component for smooth silicon etching/lithography
- Strong Base
- Wafer cleaning component
- Chemical stripper in semiconductor manufacturing
- Structure directing agent for zeolite synthesis
What is Tetramethylammonium Hydroxide (TMAH) Used For?:
- Polymers
- Semiconductor
- Zeolites
- Printed Wiring Board
- Energy Storage
- Oilfield
SACHEM is committed to the safe handling of all of our products. For more information on how to handle this product safely please visit our TMAH Safety Services page. You can also request a TMAH safety and handling presentation that includes information on toxicity, PPE suggestions and more.
Top 12 FAQs about Tetramethylammonium Hydroxide
Available Products
Synonyms: Ammonium Tetramethyl Hydroxide, TMAOH, TMAH, TMH,
Here is our store front on chembuyersguide.com: http://chembuyersguide.com/partners/sachem.html
Strategies for (TMAH) Recovery and Recycling
SACHEM has developed a closed loop recovery and recycling system for TMAH. It helps in the safe handling of TMAH by limiting exposure.
Industrial Uses
One of the industrial uses of TMAH is for the anisotropic etching of silicon.[10] It is used as a basic solvent in the development of acidic photoresist in the photolithography process, and is highly effective in stripping photoresist. TMAH has some phase transfer catalyst properties, and is used as a surfactant in the synthesis of ferrofluid, to inhibit nanoparticle aggregation.
TMAH is the most common reagent currently used in thermochemolysis, an analytical technique involving both pyrolysis and chemical derivatization of the analyte.
TMAH belongs to the family of quaternary ammonium hydroxide (QAH) solutions and is commonly used to anisotropically etch silicon. Typical etching temperatures are between 70 and 90 °C and typical concentrations are 5–25 wt% TMAH in water. (100) silicon etch rates generally increase with temperature and increasing TMAH concentration. Etched silicon (100) surface roughness decreases with increasing TMAH concentration, and smooth surfaces can be obtained with 20% TMAH solutions. Etch rates are typically in the 0.1–1 micrometer per minute range.*
